|
Remember, the solid and liquid phases of H 2O can coexist at 0☌. Solid has the least energetic state atoms in solids are. If heat is added to ice at 0☌, some of the solid changes phase to make liquid, which is also at 0☌. The three phases of matter that you frequently encounter are solid, liquid and gas (see Figure 11.8). The most commonly known phase changes are those six between solids, liquids, and gasses. 
Below is a complete list of the names of these phase changes. Condensation is when a gas changes directly to a. Matter undergoes phase changes or phase transitions from one state of matter to another. Brownian motion describes how visible particles are seen moving due to invisible molecules bumping into them. Deposition occurs when a gas changes directly to a solid. Sublimation occurs when a solid changes directly to a gas. A phase change concept map is generally a graphical illustration of the physical stages of a substance under various conditions of pressure and temperature. Melting occurs when a solid changes directly to a liquid. In other words, phase changes are isothermal (isothermal means “constant temperature”). Types of Phase Changes Freezing occurs when a liquid changes to a solid. Different smell or taste (do not taste your chemistry experiments, though!).\), the temperature of a substance does not change as the substance goes from one phase to another.Bubbles are formed (but the substance is not boiling-you made a substance that is a gas at the temperature of the beginning materials, instead of a liquid).As long as you are at 100 C, you can change the phase by changing the pressure on the system. Imagine a vertical line through this diagram- for water, choose 100 degrees C. Unexpected color changes (a substance with a different color is made, rather than just mixing the original colors together). In the video here, Sal uses a horizontal line through the phase diagram.Temperature changes (either the temperature increases or decreases). 
Observations that help to indicate chemical change include: 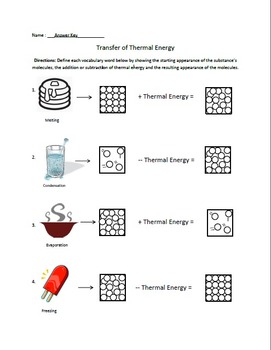
Some of the evidence for chemical change will involve the energy changes that occur in chemical changes, but some evidence involves the fact that new substances with different properties are formed in a chemical change. Points of Change CHEMISTRY TERM, PHASE CHANGE Fusion/Melting Freezing Vaporization/Boiling Condensation Sublimation Deposition, Solid to a Liquid Liquid to a. Physicists analyzing data from gold ion smashups at the Relativistic Heavy Ion Collider (RHIC), a U.S. We have to make other observations to indicate that a chemical change has happened. Each phase can directly change into the others and vice versa. We can't actually see molecules breaking and forming bonds, although that's what defines chemical changes. There are 3 normal phases of matter that we are concerned with - solid, liquid, and gas. \): Burning of wax to generate water and carbon dioxide is a chemical reaction.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
